An Internal Medicine Perspective on Safety, Science, and Consumer ConcernsThe rise of GLP-1 receptor agonists (GLP-1 RAs) — such as semaglutide (Ozempic, Wegovy), liraglutide (Saxenda), and tirzepatide (Mounjaro) — has revolutionised the management of obesity and type 2 diabetes. But with the explosion of their use has come an unexpected question from patients and cosmetic …
An Internal Medicine Perspective on Safety, Science, and Consumer Concerns
The rise of GLP-1 receptor agonists (GLP-1 RAs) — such as semaglutide (Ozempic, Wegovy), liraglutide (Saxenda), and tirzepatide (Mounjaro) — has revolutionised the management of obesity and type 2 diabetes. But with the explosion of their use has come an unexpected question from patients and cosmetic practitioners alike:
Can I safely have dermal fillers while taking a GLP-1 drug
Over the past few years, dermatologists and cosmetic physicians have reported isolated cases of allergic or inflammatory reactions occurring at dermal filler sites in patients taking GLP-1 medications. While these reports are rare, they raise legitimate questions about immune mechanisms, filler safety, and best practices for clinicians and consumers.
This article reviews what is currently known — and what remains uncertain — from the standpoint of internal medicine, integrating insights for both doctors and patients.
Background: Two booming trends collide
GLP-1 receptor agonists work by mimicking the natural incretin hormone GLP-1, stimulating insulin secretion, slowing gastric emptying, and suppressing appetite. These effects produce significant weight loss and improve metabolic control, making GLP-1 drugs among the most prescribed new medications worldwide.
At the same time, the cosmetic market has seen enormous growth in hyaluronic-acid (HA) dermal fillers, used for facial contouring, wrinkle reduction, and rejuvenation. Many GLP-1 users — especially those undergoing rapid weight loss — seek fillers to restore lost facial volume (“Ozempic face”).
Given this overlap, it’s no surprise that clinicians are now observing interactions between the two.
What has actually been reported?
The evidence so far comes mainly from case reports and small case series, rather than controlled clinical trials.
- Ouellette et al. (2023) described two patients developing dermal hypersensitivity reactions soon after starting semaglutide.
- de Oliveira Ciaramicolo et al. (2024) reported a case of delayed facial swelling occurring months after hyaluronic-acid filler injection, which worsened after the patient began Ozempic.
- Broader reviews, such as Hong et al. (2024), also document late-onset inflammatory responses in filler patients following various immune triggers — including infections, vaccines, and systemic medications.
- Moreno-Borque et al. (2024) reported a delayed-type hypersensitivity reaction linked to liraglutide.
Although these are rare, the sheer number of people now using both GLP-1 medications and dermal fillers means such reactions deserve careful attention.
Possible mechanisms — how could GLP-1 drugs trigger filler inflammation?
(a) Immune activation by peptide-based drugs
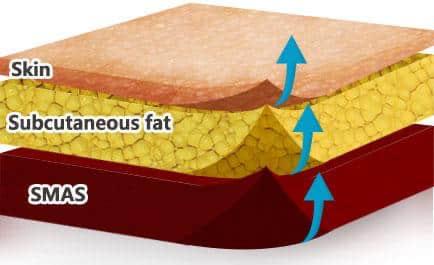
GLP-1 RAs are protein or peptide molecules capable of stimulating the immune system. A small fraction of patients develop injection-site rash, urticaria, or delayed hypersensitivity. Systemic immune activation could, in theory, reignite a low-grade foreign-body response around filler material that had previously been well tolerated.
(b) The “second-hit” phenomenon
Dermal fillers remain biologically active implants; macrophages and fibroblasts continuously remodel the injected HA. Any systemic immune stimulus — such as infection, vaccination, or new medication — can act as a “second hit,” precipitating local inflammation. A GLP-1 drug initiation or dose escalation could serve as that trigger in susceptible individuals.
(c) Tissue remodeling from rapid weight loss
GLP-1 medications often cause fat and volume loss in the face. The resulting mechanical changes may make previously placed fillers appear lumpy, displaced, or inflamed. Though this is not an “allergy,” the cosmetic changes can mimic inflammatory reactions.
These mechanisms are biologically plausible but not yet proven. No population-level data confirm causation.
What do these reactions look like clinically?
Most cases present within weeks to months of GLP-1 therapy initiation or dose change. Common features include:
- Localised facial swelling or puffiness at previous filler sites
- Tenderness, erythema, or indurated nodules
- Occasionally mild systemic malaise or low-grade fever
Symptoms may fluctuate with dosing or resolve spontaneously. The differential diagnosis includes infection, vascular occlusion, biofilm formation, or autoimmune granuloma.
From an internal-medicine standpoint, clinicians should:
- Exclude infection with basic laboratory tests if systemic symptoms are present.
- Photograph and document lesions.
- Liaise with dermatology or cosmetic colleagues for ultrasound or hyaluronidase testing.
- Consider referral if symptoms persist or diagnosis is unclear.
Read More : For more information about the effects of GLP-1 on health, please visit www.drnimaghadimi.com.
Management and treatment strategies
Treatment depends on severity and underlying cause:
- Infectious complications: Empiric antibiotics, incision and drainage if abscess is present.
- Non-infectious inflammatory reactions: Short courses of oral corticosteroids or intralesional steroids often produce rapid improvement.
- Persistent or nodular reactions: Hyaluronidase can safely dissolve HA filler, relieving inflammation.
- Refractory granulomatous inflammation: Rarely, methotrexate or biologic therapy may be indicated under specialist care.
- Medication review: Weigh the metabolic benefits of continuing GLP-1 therapy against recurrence risk. Temporary discontinuation may help in select cases, though evidence is anecdotal.
Guidance for clinicians — bridging internal medicine and cosmetic practice
Internal medicine physicians are increasingly encountering patients who combine metabolic therapy with aesthetic interventions. Effective management requires coordination and education.
Counselling points:
- Acknowledge the uncertainty: Reactions are possible but uncommon; causality remains unproven.
- Monitor and document: Encourage patients to report new or delayed swelling at filler sites.
- Timing of procedures: Some clinicians recommend avoiding filler injections during the initial titration phase of GLP-1 therapy; waiting until the dose is stable may reduce risk.
- Interdisciplinary communication: The prescribing physician and cosmetic injector should share information about the patient’s medication and filler history.
For diabetic patients, abrupt discontinuation of GLP-1 drugs is not advisable without consulting their endocrinologist. The potential metabolic benefits usually outweigh the cosmetic risks.
What should consumers know?
(a) Most people can safely have fillers on GLP-1 therapy.
The vast majority of users experience no problems at all. The reported reactions are rare and typically manageable.
(b) Understand the difference between “Ozempic face” and true allergic reaction.
Rapid fat loss can cause hollowing or skin laxity, which may be mistaken for filler changes. True allergic or inflammatory reactions involve redness, swelling, and tenderness — not merely facial thinning.
(c) Choose experienced practitioners.
Use medically qualified injectors who record the filler brand, batch number, and injection sites. This ensures proper follow-up if problems arise.
(d) Report any unusual reactions early.
Prompt review allows for early intervention with corticosteroids or hyaluronidase, preventing prolonged inflammation or fibrosis.
(e) Don’t rely on social media anecdotes.
Many online stories conflate cosmetic dissatisfaction with medical reactions. Always seek medical advice before making decisions about continuing or stopping medication
Future directions
To date, there are no formal guidelines from endocrinology or dermatology societies on managing dermal fillers in GLP-1 users. However, professional organisations such as the American Society for Dermatologic Surgery (ASDS) and the Australasian College of Cosmetic Surgery and Medicine (ACCSM) have noted the importance of pharmacovigilance and reporting such events to regulatory authorities.
Further research should focus on:
- Establishing registry-based data on filler reactions in GLP-1 users
- Identifying genetic or immunologic susceptibility factors
- Studying immune pathways modulated by GLP-1 analogues
Until more evidence emerges, a balanced, patient-centred approach — weighing metabolic health, cosmetic goals, and safety — is key.
Conclusion
At present, there is no absolute contraindication to receiving dermal fillers while on a GLP-1 receptor agonist. However, physicians and patients should remain alert to the small but real possibility of delayed inflammatory reactions. Most cases resolve with standard anti-inflammatory management and rarely necessitate cessation of GLP-1 therapy.
For the internist, awareness of these interactions reflects the expanding intersection between metabolic medicine and aesthetic practice. For the consumer, informed choice and good communication with both the prescribing doctor and injector remain the best safeguards.
Key References
- Ouellette S, et al. Dermal hypersensitivity reaction to semaglutide: two case reports. J Drugs Dermatol. 2023.
- de Oliveira Ciaramicolo N, et al. Late facial edema after lip filling with hyaluronic acid — worsening after starting Ozempic (semaglutide). J Craniofac Surg. 2024.
- Hong G W, et al. Review of the adverse effects associated with dermal fillers. Plast Reconstr Surg Glob Open. 2024.
- Moreno-Borque R, et al. Delayed type hypersensitivity reaction induced by liraglutide: case report and review. Clin Diabetes Endocrinol. 2024.
- Cedirian S, et al. Benefit-risk assessment of GLP-1 receptor agonists: cutaneous adverse events and pharmacovigilance update. Ther Adv Endocrinol Metab. 2025.
- Haykal D, et al. The role of GLP-1 agonists in aesthetic medicine. J Cosmet Dermatol. 2025.